
Food Standards Australia New Zealand (FSANZ) uses this compendium to provide best practice guidance for food regulators and the food industry.
The compendium provided microbiological criteria for ready-to-eat foods, as well as several specific food commodities: Powdered infant foods, meat products and dairy foods.
There is also expanded guidance for environmental monitoring ( swabbing) to ensure process controls are effective.
The compendium also includes information on individual pathogenic bacteria and indicator organisms.
Changes in 2022 Version:
- The latest version has a slightly different structure.
- Expansion on Dairy Products. New microbiological criteria and process control guidance for
5.a Raw Bulk Milk
5.b Butter and Dairy Blends
5.c Cheese ( Heat Treated Milk)
5.d Cheese ( Raw Milk)
5.e Dairy based Dips and Desserts
5.f Dried Milk Powder
5.g Ice cream and Frozen Products
5.h Pasteurised liquid milk and cream
5.i Powdered Infant formula
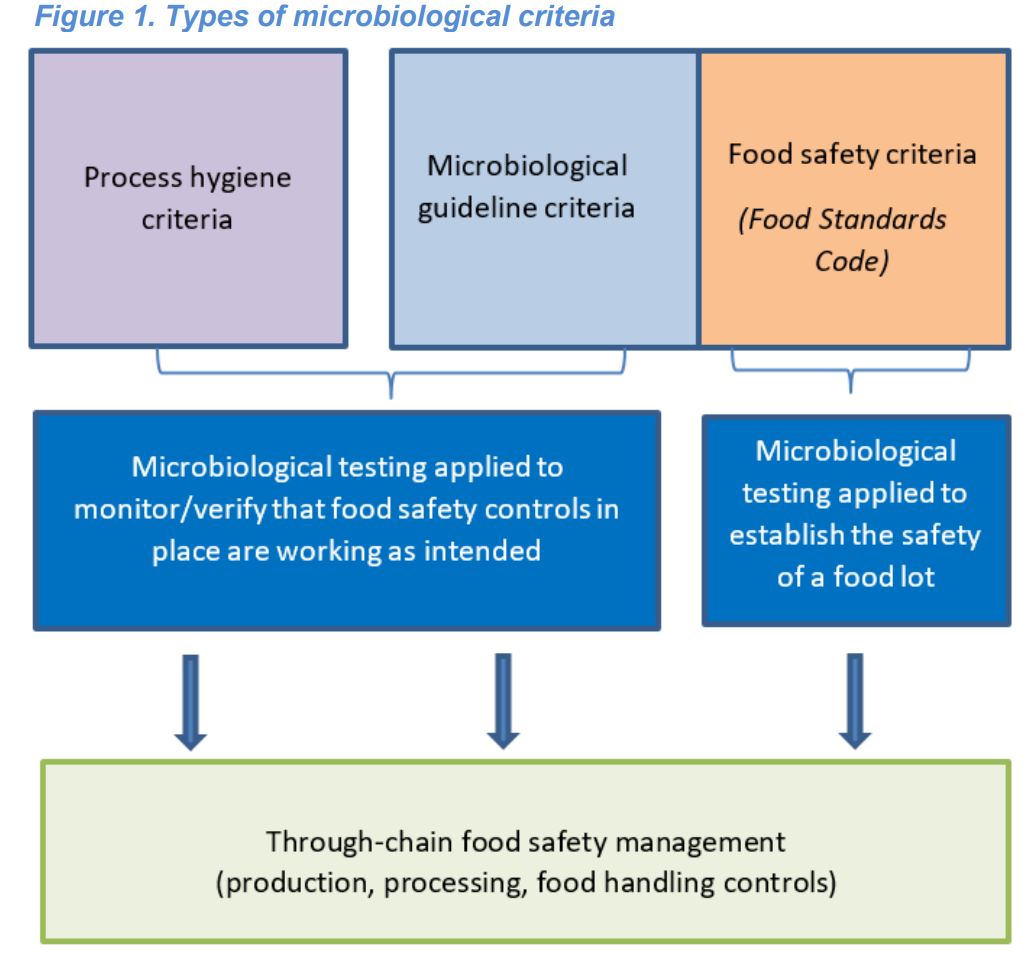
5.j Raw Goat Milk - There is additional guidance on the use of microbiological criteria, and how the compendium is used , in conjunction with the FSANZ Food Standards Code.(see below Figure 1).
- Ready to Eat Food. Pathogen testing in RTE food, Table 2.1 now in alphabetical order.
- Powdered infant formula: Adjustment of non regulatory criteria.Testing for Bacillus cereus is recommended. Process control guidance diagram. Table 3.2 Microbiological guideline criteria for powdered infant formula products
- Dairy Products.
Guidance on controls and different types of microbiological criteria.
Guidance on Dairy microbiological testing ( Section 5.2) including typical testing and sampling.
Microorganisms important in the dairy industry (Section 5.3) indicator organisms and pathogens.
Microbiological criteria for dairy product categories (Section 5.4)
Table 5.1 Microbiological criteria for raw bulk milk
Table 5.2 Process control for the production of butter and dairy blends ( Previously processed liquid milk and cream)
Table 5.3 Microbiological guideline criteria for butter and dairy blends- clearer presentation of information
Table 5.4 Process control for the production of cheese (heat-treated milk)
Table 5.5 Microbiological guideline criteria for cheese (heat-treated milk)
5.d Cheese (raw milk)
5.e Dairy-based dips and desserts
Table 5.8 Process control for the production of dairy-based dips and desserts
Table 5.9 Microbiological guideline criteria for dairy-based dips and desserts
5.f Dried milk powders
Table 5.10 Process control for the production of dried milk powders
Table 5.11 Microbiological guideline criteria for dried milk powders
5.g Fermented milk products
Table 5.12 Process control for the production of fermented milk products
Table 5.13 Microbiological guideline criteria for fermented milk products
5.h. Ice cream and other frozen products
Table 5.14 Process control for the production of ice cream and other frozen products
Table 5.15 Microbiological guideline criteria for ice cream and other frozen products
5.i Pasteurised liquid milk and cream
Table 5.16 Process control for the production of pasteurised liquid milk and cream
Table 5.17 Microbiological guideline criteria for pasteurised milk and cream
5.5 Corrective Actions
Table 5.18 Recommended corrective actions (if failure of microbiological guideline criteria)
Table 5.19 Recommended corrective actions (if failure of process hygiene criteria)
5.6 Clearance program- guidance and sampling
5.7 Environmental monitoring - guidance. - 6.1 Environmental Monitoring- Introduction
6.2 A typical monitoring process
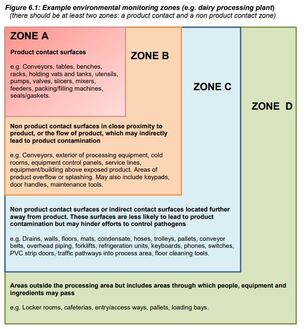
Figure 6.1: Example environmental monitoring zones- expansion of 3 zones to now 4 zones (A-D).
See Figure 6.1 below.
6.3 Recommended corrective actions- for the 4 zones A-D.
6.4 Monitoring for Listeria monocytogenes- specific guidance - Appendix 1 Pathogens.
Clostridium botulinum (added)
Shigella (added) - Appendix 2 Indicator Organisms ( now alphabetical order: Coliforms, Enterobacteriaceae, Escherichia coli, Listeria spp, Standard plate count (SPC)
 RSS Feed
RSS Feed